Entropy is a fundamental concept in physics and chemistry that plays a critical role in shaping the natural world. At its core, entropy is a measure of the disorder or randomness of a system. It tells us how much energy is unavailable to do useful work and how much energy is lost to heat.
The concept of entropy was first introduced by Rudolf Clausius in the mid-19th century as part of his work on thermodynamics. Clausius defined entropy as a measure of the amount of heat that cannot be converted into work, and he established the second law of thermodynamics, which states that the entropy of any isolated system always increases over time.
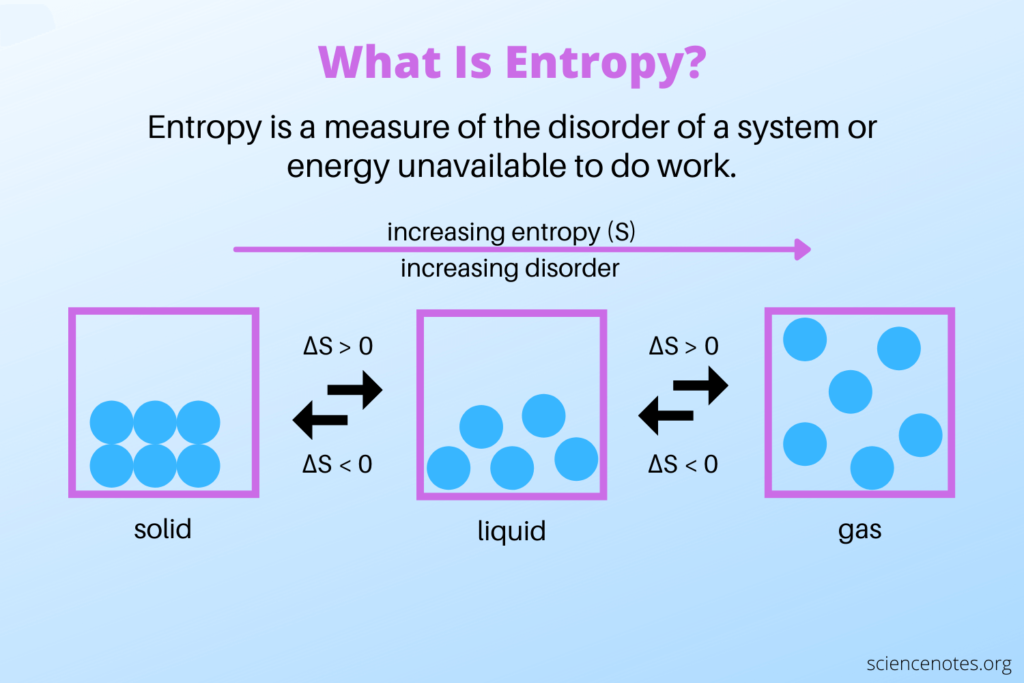
One of the most famous examples of entropy in action is the behaviour of a gas in a closed container. When a gas is confined to a container, its molecules bounce around randomly, creating a state of disorder. As time goes on, the gas will continue to expand until it reaches a state of maximum entropy, at which point the molecules are evenly distributed throughout the container.
Entropy also plays a critical role in chemical reactions. The process of chemical reactions involves the breaking and formation of chemical bonds, which can either increase or decrease entropy depending on the nature of the reaction. For example, combustion reactions, such as the burning of fuel, typically release heat and increase the disorder of the system, leading to a higher entropy.
Entropy is not just a concept that applies to physics and chemistry, but it also has applications in other fields, including information theory and biology. In information theory, entropy is used to measure the amount of uncertainty or randomness in a message or data set. In biology, entropy is used to study the flow of energy and the organization of living systems.
The concept of entropy has also led to important technological advances, such as the development of heat engines and refrigeration systems. By understanding how entropy works, scientists and engineers have been able to design more efficient and effective systems for energy conversion and storage.
In conclusion, entropy is a fundamental concept that plays a critical role in shaping the natural world. It is a measure of the disorder or randomness of a system and is a key concept in fields ranging from physics and chemistry to information theory and biology. By understanding entropy, we can better understand the world around us and develop more efficient and effective technologies to harness its power.









